Cooking up volts in the galley

Issue 105 : Nov/Dec 2015
Jones was a large man who said everything with a smile, even if he was insulting you. As I walked into the marina’s restaurant and bar, a favorite hangout for boaters, I heard him blurt out with a volume that echoed off the walls, “That’s crap! It can’t be done.”
Scott was a quieter, softer-spoken kind of guy who replied simply, “Hey, I’m just telling you what I saw on the Discovery Channel last night.”
Jones noticed me and, without a hello or greeting of any kind, stated: “Doc, help us settle an argument.”
It was the quiet time for restaurants; the lunch crowd had departed and the dinner crowd had yet to arrive. I found a comfortable bar stool three seats down from Jones and Scott. Sally, the barmaid, wore her typical Hooters-inspired outfit and instinctively grabbed a cold mug and started filling it with draft beer.
I asked her, “What’s the debate today?”
With a thick southern accent that suited her personality well, she replied, “Something about batteries.”
Jones looked at me and got back to his original thought, “Did Thomas Edison have an electric boat that could travel 70 miles on a single charge?”
I finished a sip of beer and replied, “It’s true and, if I remember correctly, it could carry about 20 people.”
I continued with a bit more information, “Batteries were different back in the early 1900s: different materials, different acids. Most of them were what they call ‘Primary Batteries.’ After they’d go dead you’d throw them away, like your typical AA or AAA batteries.”
Scott was curious. “They weren’t lead-acid batteries like in my car?”
I responded, “Nope, I forget what these were made from, but you can make a battery out of most dissimilar metals. I saw a guy on YouTube make a battery out of a lemon, a piece of copper, and some steel.”
Jones pounced on my comment. “Are you saying you can make a battery?”
The conversation seemed to have turned into an inquisition. “I think so. I could probably make one that can power up a handheld GPS or an LED flashlight or maybe one that can even create a spark to start a fire.”
Jones gave a snort of disbelief before giving his less than eloquent response: “Bull****.”

The challenge
The large windows that usually provide a picturesque view of the marina showed dark clouds on the horizon and rain began to fall. I made myself comfortable on my bar stool, took a sip of beer, and replied, “What’s the bet?”
Jones put some thought into it, “OK, MacGyver, you’ve got to make a battery using only stuff that can be found on a typical sailboat or you owe me a beer.”
I liked the challenge, but I wanted a little entertainment for my efforts, “Can I attach it to your tongue when I’m done?”
He pondered the proposition for a quick second and responded, “Sure, but when I don’t flinch you’re gonna owe me two beers.”
I addressed everybody who was listening, “I will need 20 pennies, aluminum foil, salt, vinegar, cardboard, scissors, and duct tape.”
Everybody looked at Sally who, without having to be asked, went about collecting what I needed.
They all watched me closely. I poured vinegar and salt into a bowl and tossed in all of the pennies. Answering the raised eyebrows I explained, “It’s to remove the clear coating and expose the copper.”
I began cutting out small discs of cardboard and aluminum foil the same size as the pennies, 20 each. I tossed the cardboard discs into the vinegar too.
Scott asked, “What’s the plan here, Doc?”
I continued working as I responded, “I will sandwich a piece of vinegar-soaked cardboard between a penny and a piece of aluminum foil and that will create a battery. Most batteries are a form of rusting. When steel turns into rust, it loses electrons. My aluminum here is going to do its form of rusting and lose electrons.”
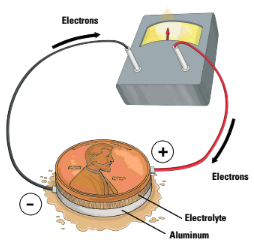
Jones looked at my small battery, chuckled and remarked, “Hurry up, Doc, I’m thirsty for a free beer.”
I continued, “But electrons have a very strong repulsion to one another like . . .”
Before I finished my sentence, Scott chimed in with a big smile, “Like the female race has to our friend Jones here.”
Jones gave Scott a quick disapproving glance as I went on, “These electrons will push against each other like bubbles in a soft drink. When you unscrew the cap of a Coke you hear the bubbles being forced out. Turning on an electrical switch is like unscrewing the cap on a soft drink except you let the electrons escape. When we attach this to Jones, the electrons should be pushed from the aluminum, through his tongue and to the copper, giving him a good jolt.”

Jones looked a bit worried as I created 20 of the small batteries and then stacked them together, one on top of the other like batteries in a flashlight. I used duct tape to hold the whole contraption together and rolled aluminum foil into wires to help connect the battery to Jones’ tongue.
With my battery complete, I asked Sally for a pinch of steel wool. When Jones began to protest the additional material I said, “It’s just for a test.”
I touched the two homemade wires to a cotton ball-sized clump of steel wool and — like magic — it began to glow red. When I touched a piece torn off a napkin to it, the paper caught on fire.

Jones looked apprehensively at the burning steel wool in the ashtray, but everybody else was grinning. Sally was almost giddy with delight and addressed Jones, “All right, Big Boy, get your head down here to the bar so we can attach this thing to your tongue.”
Jones was more mellow than usual and raised his hands as if he were at gunpoint, “OK guys, I concede.”
Sally became our spokesperson and in a sharp, but friendly, tone replied, “That wasn’t the bet. Get on down here or be prepared to be ridiculed for the rest of your life.”
Jones slowly turned his hat around backward and lowered his chin to the bar. Sally moved in closely with the homemade battery. A couple of the regulars got out of their seats and stood next to Sally to watch the show.
The rain had stopped. As I headed toward the door I heard a strange yelp followed by a lot of laughter.
Just as I closed the door, I heard someone holler, “Shock him again!”
Conrad Cooper is a published author (Own Less & Live More) who enjoys writing about boats and the cruising lifestyle. Conrad and his wife, Roxanne, are currently refurbishing their third boat and cruising the Gulf Coast. Visit their blog at www.LifeOfLatitude.com.
Thank you to Sailrite Enterprises, Inc., for providing free access to back issues of Good Old Boat through intellectual property rights. Sailrite.com












