E10 gasoline is now a fact of life, but its devils don’t have to be
Why is ethanol problematic for marine engines but not for cars? The only noticeable difference for the family car is reduced gas mileage, the result of ethanol-blended fuel containing less energy per gallon. The marine problems are different and far more severe, and stem from moisture, irregular usage, and smaller scale.
Small engines have always been a tribulation. The high surface-to-volume ratios of their small tanks accelerate oxidation and gum formation and evaporate the volatile components that give gasoline its starting ginger. Smaller carburetor passages are more clog-prone, and simple engines lack the sensors and computers car engines use to optimize the mixture. Many small engines aboard boats are used only sporadically, further increasing the potential for corrosion and stale fuel.
More than 90 percent of the fuel market is now 10 percent ethanol, or E10. We worry that E15 is knocking on the door, likely to become a pervasive reality in the next decade. It’s no longer an issue of lobbying for this scourge to go away, it’s a matter of learning to live well with the fuel we have.
Moisture seldom finds its way into the automotive tank because the tank is sealed. Venting, when it occurs, is through a carbon canister, which removes most of the water. The canister is then purged clean when the car runs. Per US Coast Guard regulations, boat tanks cannot be sealed in any way that might increase pressure. Even slight pressurization increases the risk of leakage and subsequent explosion. If a car develops a minor leak, the fuel will probably just drip on the ground, and at the very worst, you can get out and walk away. A dripping tank aboard a boat can lead to gasoline fumes in the bilge, and the results can be catastrophic.
Cars are used daily, while a boat that is used 20 to 30 weekends each year is considered very busy indeed. And while gasoline will seldom stay in the family car for more than two weeks, it might sit in an outboard-motor tank for months at a time. Indeed, as sailors, we take pride in running the engine for as few minutes as it takes to clear the jetty, which is one of the worst things we can do for the engine. Any gas that remains in the tank will sit through the winter, evaporating, drawing moisture, and polymerizing.
Finally, there is the matter of size. The fuel passages in the kicker on your dinghy, or even an Atomic 4, are logically 5 to 30 times smaller in cross-sectional area than in the first car you drove, back when cars had floats and jets rather than fuel injection. It does not take much to plug these passages enough to restrict the flow, throw off the mixture, and strand you somewhere.
All of these problems with E10 are quite solvable through better practices, additives, and minor equipment modifications. I have fought with these problems, but perhaps being a chemical engineer in the refining business gave me a leg up. It is often said about diesel engines that the key to reliable operation is clean fuel. The same is true for gasoline, although the path is different.

Drew tested evaporation water absorption by filling 1-liter jars with 500 ml of E10 and allowing them to breathe for 2 months, either freely or through a scale-size silica-gel or carbon filter. The silica-gel filter consistently showed the lowest evaporation rates because it was less affected by moisture.
Keeping the fuel dry
Gasoline and water don’t mix. Traditional gasoline would not draw water from the air. If a few drops of water found their way into the gas tank, they would sink to the bottom and do no harm other than eventually rusting a hole in a steel tank. E10 is different.
Ethanol is soluble in gasoline, but add just 0.5 percent water and all that water will be drawn to the alcohol, and a phase containing the water and all of the ethanol will fall to the bottom. Because the solubility of water in E10 changes with temperature, fuel that was stable during the summer, collecting a few drips and drawing water from the air, can suddenly become unstable when the temperature drops from the 80s and 90s of summer to the 40s and 50s of fall with the first cold snap. This is more of a problem with smaller tanks because they cool more quickly.

The coupons he used in his corrosion tests included aluminum, brass, copper, and steel in galvanically coupled groups. The corrosion on the aluminum one is similar to that in the carburetor bowl.
There are additives whose makers claim will prevent phase separation. Don’t use them. Testing shows this is not generally true, and on the balance, that is a good thing. Do you want to run water, perhaps including seawater, through the engine? A few additives are able to accomplish this trick, but it is, in the end, a dirty trick. They can keep the water dissolved only by means of high levels of co-solvents like butyl cello- solve or with high levels of surfactants. These are not harmless and the engine manufacturers have been working on standards that would ban this approach due to problems with intake-valve deposits.
The primary water source will always be a leaky filler cap. The cap on my first boat was located right in a drainage path, and I was constantly battling water leakage. It was only a few drops, so I installed a small separating filter (Raycor 110 series) and drew off a few teaspoons per year. That lasted until E10 arrived, after which that tiny amount of water would trigger the separation of gallons of ethanol/water mixture seemingly overnight. To get to the root of the problem, I moved the filler to a more elevated position in a location that would not allow vapors to enter the cabin or belowdeck areas. I kept the O-ring fresh and lubed it with a smear of waterproof grease a few times a year.

Pitting in this carburetor bowl was the result of mixing ethanol, brass, and aluminum.
Another potential water source is the tank’s vent. Portable tanks and integral tanks have a poor record of keeping out rain and heavy spray when the vents are open, as they must be during operation. More than once, an overnight rain (it doesn’t take much) has found me pulling lines, replacing the filter, and draining a bit from the tank. Cover the tank to prevent rain or spray from hitting the vent when it’s in use and close it when it’s not in use. Driving rain can enter a stanchion vent. Transom vents can be slapped and water driven into the tank; the best protection is a high loop in the vent hose, at least 6 inches higher than the inlet. Another solution is a P-trap vent, available from Attwood and others.
Even a few humid evenings can introduce enough water to cause trouble in the 1-liter tank of a dinghy outboard. In the case of portable tanks and integral tanks, the vent should be closed anytime the engine is not operating. If you don’t close it every time, night will come, you will forget, and it will probably rain.
Fixed tanks can benefit from adsorbent vent dryers. The EPA tested this in the process of developing evaporative emissions standards for boats (primarily, these apply only to power boats, due to tank size and production-volume limits). The unfortunate error was that the test lab used automotive test conditions, not the heavy-dew situation encountered 18 inches off the water, thus missing key design variables entirely. I set about testing tank vents, both in the lab and in the field, under more realistic high-humidity conditions.
When field testing on a number of boats in the water, we saw that the carbon in the vent filter would often be soaking wet with dew. Remember how gasoline and water don’t mix? Carbon that is wet can’t absorb gasoline fumes, because it is as though each granule is sealed in a plastic bag. Silica gel, on the other hand, while not as effective an adsorbent for hydrocarbon vapors, is not adversely affected by water vapor or fog and performs more consistently. At both laboratory-scale and in on-boat testing, silica gel outperforms carbon in keeping water out of a tank and in reducing evaporation.
I also learned that the daily out- breathing of E10 has an interesting self-regeneration effect on silica gel: Each time the ethanol breathes out, it displaces some of the water, regenerating the filter for the next cycle. As a result, the silica gel remains effective for up to 5 years, rather than the 6 months to a year manufacturers may suggest. Even spent media traps dew until it can evaporate in the morning, and it reduces oxidation and evaporation by reducing air exchange in the tank due to convection (air currents). Gas in a portable tank or an integral tank will evaporate less and absorb less water when the vent is kept closed.
I installed a vent filter on my PDQ 32 catamaran and those of several friends 8 years ago, in climates as varied as Chesapeake Bay, South Florida, and Seattle. Typically, after 5 years they required regeneration. All of the testers regenerated the gel by heating it in a pan, and none reported any difficulty. As the gel dried, it returned to its original blue color, and after cooling was ready for another 5 years of service. In my case, during those 5 years, it kept several pounds of water out of my fuel, which is important far beyond the problem of phase separation. As scary as it sounds, phase separation is not the main problem with water.

After 2 months, the best-per- forming anti-corrosion additive left the gasoline clear and the test coupons corrosion-free.
Anti-corrosion additives
Corrosion is the real problem. Additive makers claim improved stability and reduced gum formation. However, during several rounds of extended aging testing, the only thing that correlated strongly to gum formation was corrosion of copper and zinc alloys. These metal ions, once dissolved in gasoline, serve as aggressive catalysts for oxidation and polymerization. Before ethanol, corrosion rates were low, because water is insoluble in gasoline, gasoline doesn’t conduct electricity, and the ions were not mobile. But the addition of ethanol changed all that. E10 gasoline absorbs water, water supports galvanic corrosion within the fuel system (carbs contain brass jets in an aluminum body — an excellent recipe for aluminum pitting), and copper and zinc ions are now mobile and aggressive.

To test the anti-corrosion properties of the additives, I placed standard ASTM corrosion coupons in vented bottles for 2 months, along with 150 PPM (parts per million) of seawater, because a wee splash into the vent is not unheard of, to compress years of corrosion into a reasonable testing period. They were galvanically coupled in groups as they might be in a fuel system; copper and brass to aluminum, steel to brass, and so forth. The bottles were fitted with a hose sized to simulate normal venting of a 10- to 20-gallon tank without allowing excessive evaporation or oxidation. They were stored in the dark, and at the end of the period, observed for relative corrosion.

In another sample, there was corrosion on the coupons and the gasoline was visibly darkened by oxidation and sludge formation.
Of the top performers, Biobor EB is my personal favorite — it stopped corrosion dead in its tracks. Mercury Quickstor, SeaFoam Motor Treatment, and ValvTect Ethanol Gasoline Treatment are also excellent. Beware of the rest; some do little to arrest corrosion, and a few can actually make it worse.
Filtration

A lawnmower fuel filter fitted to a 3.5-horsepower Mercury outboard reduced carburetor clogging significantly.
A little supplemental filtration can go a long way toward reducing on-engine filter plugging and carburetor problems. The micron rating is probably not that critical. Particles smaller than 10 microns might cause wear in a diesel injection pump, but they won’t be noticed in a carburetor. Most water-blocking elements are rated at 10 microns and I’ve had good experience with these. I have also used 2-micron elements and didn’t notice any real difference.

With no anti-corrosion additive present, venting the tank allows in moisture, which leads to corrosion.
Is a filter with a water drain required, such as used for diesel fuel? I’ve had both, and absent a chronic leakage problem (like my first boat — fix the problem), water will never accumulate in the drain, not in 10 years, because unless the gasoline phase separates in a major way, fresh E10 gas will continuously dissolve all traces of water. Should a droplet somehow condense in the tank and find its way to the filter, it might settle to the bottom, but it will quickly redissolve.

Sealing the tank to keep moisture out helps prevent corrosion.
Another problem is that the separated phase formed in E10 is not water but actually 90 to 95 percent ethanol. It does not bead up readily on the surface of the filter because its surface tension is much lower than that of water and not so different from gasoline’s. It does not readily run off the filter surface and down to the bowl because it is not as heavy as water and not much heavier than gasoline. As a result, separating filters just don’t work as well with E10 as they do with conventional gasoline or diesel. There is no harm in using a filter with a drain, just don’t be disappointed if there is nothing to drain in the normal course of sailing, and don’t expect it to help that much if the gas separates; it might help, but it might not. I’ve had both experiences.

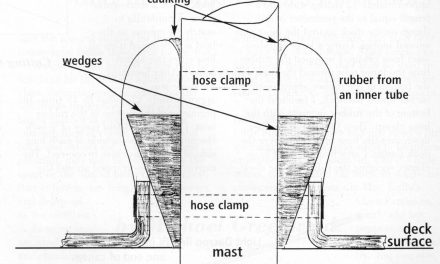
Drew installed an H2Out AVD-2 silica-gel vent filter in a high loop, at left. He also modified a Vetus no-smell vent filter to hold silica gel, at right.
This is not to say a few water droplets aren’t a problem and that a sufficiently large filter can’t help. Phase separation takes time, and often just a tiny bit of emulsion forms at the bottom of the tank. The tiny in-line filter under the outboard engine cowl is easily blocked by just a few drops of water or a little bit of this emulsion. The filter won’t look plugged and you can even blow through it, but the medium is designed to repel water. With even the thinnest coating, it becomes gasoline-repellent, much as water-repellent treatment can make canvas water- resistant. The canvas is not truly sealed, just coated with enough hydrocarbon to make the water bead up. Apply the slightest pressure — often just the touch of a finger — and the fabric will begin to leak. But this thin coating of water on a tiny factory filter can be enough to starve the engine.
I recommend a filter rated for 35 to 90 gallons per hour, even for an engine that can only burn a gallon or less. This gives the water the time it needs to either flow off or redissolve. If dirt is present, the filter will take more than 5 years to become fouled instead of once or twice each season, because the larger filter has far more capacity to hold dirt, and because the greater surface area allows it to function while partially blocked. Should you encounter a small slug of dirt from a dirty jerrycan or a few drops of water from a splash, the larger filter can handle this without stranding you. My favorites? I like the Raycor Snap, the Raycor 110 series, the Raycor 320 series, and the Sierra spin-on kits. Some small outboards — my old 3.5-horsepower Mercury two-stroke comes to mind — might have only a fine strainer in the tank. Add a lawn-mower filter under the cowl for just a few dollars and enjoy the reduction in carburetor clogging. It may clog if you leave the vent open through an overnight rain, but changing it is the work of a few minutes.
Enleanment
There is one more problem we can’t do much about: enleanment.
Gasoline with ethanol requires a slightly richer mix, but simple carburetors cannot compensate. What this means is that the carburetor will be running a little on the lean side and will be more sensitive to clogging, as it’s already getting less gas than it was designed for. This will get worse with E15, which is also thought to be damaging to seals and intake valves of engines not designed for it. The EPA does not appear to be widening approvals, and no marine engine manufacturer approves E15, so don’t use it in your boat.
As the US fuel pool moves slowly, inexorably, to all ethanol-blended fuels, newer engines have been rejetted to compensate. But expect older motors to run a little lean on E15. Because small engines are not subject to quite the same emission standards as their computerized counterparts in cars, they are designed to run the mixture a little richer than strictly required, and so far, this has been enough. Let’s hope that another 5 percent won’t be fatal.
I can proudly say I’ve learned to live in harmony with E10. I check my filler O-rings. Where I have a fixed tank, I use a silica-gel vent dryer to keep the fuel bone-dry. If the tank is portable or integral to the motor, I close it faithfully after use. I add Biobor EB as directed to prevent carburetor corrosion and gum formation. I add supplemental filters to catch anything the engine filter missed and to deal with the few drops of water that might fight their way through on occasion. The result is super-clean fuel, and the motors like that. Years ago, I learned to clean a carb under way with one eye closed and the boat bouncing enthusiastically, knowing I needed to get this sorted out before I reached the marina. Now the engines start on the first or second pull, every time, for many years at a time. That’s nice.
Portable-Tank Advice
Most of the time, a portable tank works just fine left in the cockpit and exposed to the elements. But you can do a little better.
Keep it out of the sun. E10 has considerably higher vapor pressure than conventional gasoline, resulting in tanks that bulge alarmingly in the sun. Sometimes fittings leak. Sometimes gas is forced past the needle valve into the engine (consider installing an in-line cutoff valve). The UV is not good for the tank and it isn’t good for the gasoline inside the tank.
Close the vent when the tank is not actively in use. I’ve had both integral tanks and portable tanks phase separate as a result of water that splashed in during heavy downpours in a matter of an hour.They also draw water vapor from the air and evaporate valuable volatiles into the air.
Use caution when venting. A significant puff of vapor can be released when a bloated tank is vented. Make sure to open the vent only when in a safe area away from ignition sources and located such that the fumes cannot get belowdecks.
If filling a portable tank at a fuel dock, remove it from the boat and fill it on the dock. Static and fumes are your concerns.
Living Ethanol-Free

By Cliff Moore, a Good Old Boat contributing editor.
A few years ago, I replaced the two-stroke outboard motor on my dinghy with a 2.3-horsepower four-stroke Honda, which behaved flawlessly for a very long time. Each fall, I changed the oil in the head and lower unit and, knowing that fuel goes bad over time, I drained the fuel tank.
Then something happened. Three years ago, the engine ran rough all summer. It would start, then run for a minute, then quit. Even with new fuel it misbehaved. The next summer it worked perfectly well.Then, last summer, it was back to its old tricks again, running rough and quitting. It always restarted, but it was a nuisance.
I finally figured out the problem — and the solution. The problem is the ethanol contained in gasoline sold at highway gas stations, where many trailer-boaters fill up. Ethanol readily absorbs moisture — a couple of drops through the cap vent and the engine’s not happy. While there are fuel-treatment products that are promoted as mitigating the negative effects of ethanol in gasoline, I’ve found the best solution is to simply burn only ethanol-free gas. How? The website pure-gas.org lists every service station (including marinas) in the US and Canada that sells ethanol-free gas.
I was not surprised to find that in New Jersey, where I lived until February of last year, only 10 gas stations in the entire state sold ethanol-free gas. But here in North Carolina, where I now live, hundreds are listed by town. At least three are within a mile of my home, and another couple are on the way to the marina I now sail out of.